Looking for help?
Common queries
Send us feedback
We'd love to hear your thoughts on features, usability, or what you love about Ideagen Luminate.
CAPA in manufacturing

![]() Aaron Hoare
Aaron Hoare
In the realm of quality management systems (QMS), managing Corrective and Preventive Actions (CAPAs) in manufacturing takes center stage. A strong grip on your CAPAs not only shields your QMS and organization from serious examination, but it also acts as the cornerstone of continuous quality improvement.
Key advantages of strong manufacturing CAPA processes include reduced defects, reworks, and out-of-specs; easier adherence to quality standards and regulatory demands; improved efficiency across your organization; better safety standards for end-users; significant reduction in costs.
Put simply, CAPA management in manufacturing ensures a process for getting things right first time and, crucially, making sure things stay that way. This is a must for any quality team.
If your current CAPA system seems wanting, continue reading this introductory guide to CAPA management. As an alternative, download our more in-depth guide to CAPA in manufacturing below.
How to structure a manufacturing CAPA system? Make use of the 4 ‘I’s
An effective approach to manufacturing CAPA management is to adopt a closed-loop methodology. It is up to you how many steps should be involved in your CAPA system, and this can and should vary based on factors like the size of your organization or the complexity of your manufacturing processes.
In any case, you should look to adopt a PDCA framework. The PDCA framework is proven to be effective for CAPA in manufacturing systems. In brief, the 4 ‘I’s guide the implementation of a robust CAPA strategy in manufacturing. For a deeper dive into the 4 ‘I’s, refer to our CAPA in manufacturing whitepaper.
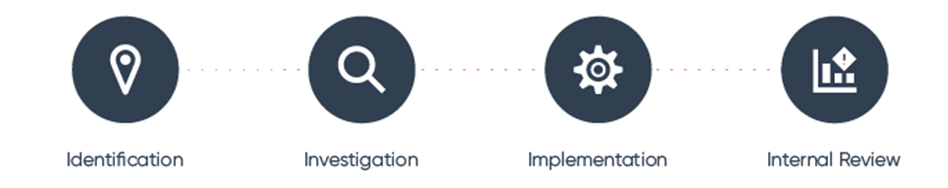
Stage 1 – Identification of non-conformities
The most important facet of your CAPA plan involves identifying and assessing non-conformances. Failure to do so can be detrimental to your manufacturing operation, as recalls and regulatory fines can quickly snowball into a significant financial burden. This is why having strong manufacturing CAPA processes is so important.
Regular operational checks and reviews should be conducted to unearth non-conformances for CAPA management.
Other sources of identification might be regulatory inspections, third-party audits, and customer feedback. Ensure you have an organized system to capture and evaluate these inputs. Make sure you take your time to collect as much information as possible, otherwise you may find yourself reacting to situations but missing the bigger picture of what the real issue is.
Stage 2 – Carry out an investigation
After a non-conformance has been found and its impact measured, a thorough root cause investigation is required. Root cause analysis should always form the core of all CAPA in manufacturing processes.
Quality tools, techniques, and a cross-functional team should be utilized for the investigation. The goal here is to examine all circumstances related to the root of the non-conformance. We go into more detail on the tools you can use for root cause analysis in our in-depth whitepaper.
Stage 3 – Implement effective actions
Discovering your root causes will aid in formulating a list of actions to eliminate the non-conformance and prevent its reoccurrence. When creating your CAPA action plan, clearly set out each action and task, and any required changes to your processes, procedures, or information streams.
Make sure anyone involved has the proper training and competency to implement these actions effectively. Finally, you want to make sure you are measuring and documenting the outcomes from your changes, as well as documenting the rationale behind the changes.
Stage 4 – conduct an internal review
Internal audits are a crucial component of your manufacturing CAPA system. As part of these audits, you should answer several questions:
-
Has the real root cause of the nonconformity been identified, not just a symptom of a deeper problem?
-
Have you seen a reoccurrence of the nonconformity, and if so, is it an isolated incident?
-
Have the actions taken had any unexpected effects on other parts of your organization?
-
Has your corrective and preventive action plan been successful?
This leads us to the 4th ‘I’ – the internal review. It's important to continuously assess the effectiveness of your CAPAs before closing them off.
Strengthen your manufacturing CAPA management system
Are you struggling with managing numerous CAPAs all at once? Having issues synchronizing your quality processes? Worried about overlooking serious non-conformances?
It might be beneficial to explore a specialized CAPA software solution, such as Ideagen Quality Management. Ideagen QMS platform integrates robust data gathering and reporting, thereby simplifying the identification of non-conformances.
It also boasts advanced intelligence dashboards to reveal quality discrepancies and their root causes. And, with automated and digitally enabled workflows, tasks can be assigned to the right team members when they're needed, ensuring comprehensive visibility of your manufacturing CAPA management. If you're keen to enhance and streamline your manufacturing CAPA procedures, discover more about Ideagen QMS and arrange a demo with our experts today.

Coming from Skills? Create an Ideagen Luminate account to link your learner profile. Learn more.

Coming from Skills? Create an Ideagen Luminate account to link your learner profile. Learn more.

Coming from Skills? Create an Ideagen Luminate account to link your learner profile. Learn more.

Coming from Skills? Create an Ideagen Luminate account to link your learner profile. Learn more.
Login with SSO
Ideagen Employee Login Login with Ideagen HubEnter your E-mail address. We'll send you an e-mail with instructions to reset your password.


